 
The electronic configuration of Chlorine will be 1s2 2s2 2p6 3s2 3p5. How do you write the electron configuration for Chlorine? 
What is the electronic configuration of Chlorine 17? What is the boiling Point of Chlorine in Kelvin?īoiling Point of Chlorine in Kelvin is 239.11 K. Melting Point of Chlorine in Kelvin is 171.6 K. What is the melting Point of Chlorine in Kelvin? By adding one more electron we get a negatively charged Cl- ion with a net. What is the boiling Point of Chlorine?īoiling Point of Chlorine is 239.11 K. A neutral chlorine atom, for example, contains 17 protons and 17 electrons. Chlorine has 17 electrons out of which 5 valence electrons are present in the 3s2 3p5 outer orbitals of atom. How many valence electrons does a Chlorine atom have?Ĭhlorine has 5 valence electrons. Chlorine has an atomic number of 17 and an atomic mass of 35.45, meaning that an atom of chlorine consists of 17 protons, 17 electrons, and 18 neutrons. The element Chlorine was discovered by W. What is the color of Chlorine?Ĭhlorine is of Yellow color. It is located in group 17 and period 3 in the modern periodic table. 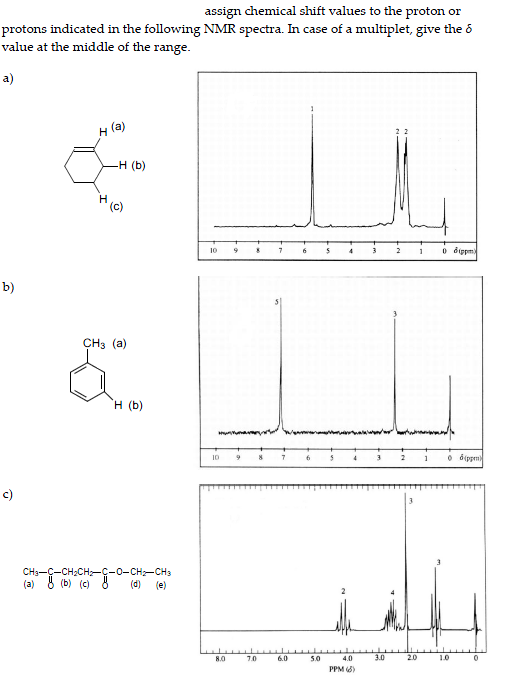
Chlorine is the 17 element on the periodic table. What is the position of Chlorine in the Periodic Table?Ĭhlorine is a chemical element with the symbol Cl and atomic number 17. Chlorine is a chemical element with symbol Cl and atomic number 17. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. Two atoms with the same number of protons and different number of neutrons. You are told that the mass number is 79, and so there are a total of 79 protons plus neutrons. 
Elements from Groups 1 and 17 can combine to form ionic compounds in a one-to-one ratio. While a chlorine atom always has 17 protons, it can have any number of neutrons. Bromine has an atomic number of 35, and so has 35 protons. The abbreviated electronic configuration of Chlorine is 3s2 3p5. Note that not all of the protons and neutrons are visible. What is the abbreviated electronic configuration of Chlorine? Math Processing Error Math Processing Error Math Processing Error Math Processing Error Math Processing Error Explanation: Based on the periodic table, the atomic number ( Math Processing Error of chlorine is 17. The electronic configuration of Chlorine is 1s2 2s2 2p6 3s2 3p5. By convention, elements are organized in the periodic table, a structure that captures important patterns in their behavior.Devised by Russian chemist Dmitri Mendeleev (18341907) in 1869, the table places elements into columnsgroupsand rowsperiodsthat share certain properties. For example, oxygen has an atomic number of. The basic particle that constitutes a chemical element is the atom, and each chemical element is distinguished by the number of protons in the nuclei of its atoms, known as its atomic number. Now imagine that you could somehow keep dividing the speck of carbon into smaller and smaller pieces.What is the electronic configuration of Chlorine? A chemical element is a chemical substance that cannot be broken down into other substances. Each speck of carbon would still have all of the physical and chemical properties of carbon. Imagine taking a small piece of carbon and grinding it until it is a fine dust. Remember the atomic number of an element. The graphite in your pencil is composed of the element carbon, a nonmetal. What is the mass number of the isotope with the symbol 37/17 Cl a) 17 b) 18 c) 35. Since we know the atomic number of Cl is 17, we can work out that Cl-35 has 18 neutrons, and Cl-37 has 20 neutrons. Carbon-13 behaves chemically in exactly the same way as the more plentiful carbon-12. Chemical behavior is governed by the number of electrons and the number of protons. While the presence of isotopes affects the mass of an atom, it does not affect its chemical reactivity. The atomic number of each element increases by one, reading from left to right. Period A horizontal row in the periodic table. Members of a group typically have similar properties and electron configurations in their outer shell. The nucleus of a given carbon atom will be one of the three possible isotopes discussed above. 17 35.45 Glossary Group A vertical column in the periodic table. Tin has ten stable isotopes, which is the most of any known element. Carbon has only three natural isotopes, but some heavier elements have many more. Naturally occurring samples of most elements are mixtures of isotopes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
